Antigen tests measure fungal cell wall molecules (mainly carbohydrates) that are shed into the body fluids. Titre will depend on species/isolate, fungal load, degree of angioinvasion, clearance rate from blood and whether the patient is already taking antifungals. Therefore, it is vital to understand the performance of each test within a given patient group, as well as likely sources of false positive and negative results.
Both the cryptococcal antigen (CrAg) test and the Histoplasma antigen test are included on the 2nd edition of the WHO Essential Diagnostics List for use in diagnosing invasive infections in patients with HIV.

How to perform & interpret tests

Interpretation of results
As with all biomarkers, care must be taken to interpreting results in the context of the patient’s overall clinical presentation, immune status, sample type and any other mycological evidence available – please refer to the individual clinical guidelines for each disease. For example, a positive serum galactomannan result could support a diagnosis of probable invasive aspergillosis, but direct mycological evidence (microscopy or culture from a sterile site) would be required for proven invasive aspergillosis.
Please be aware of sources of false positives/negatives and the performance parameters (PPV, NPV, etc) for your patient population when interpreting results.
Fungal antigens are generally poorly immunogenic and so not usually heavily complexed with specific antibody, with the exception of the Candida mannan antigen. However, they may be cleared from the blood in neutropaenic patients.

Point-of-care testing
Several antigen tests are now available as point-of-care (AKA bedside) tests, with a dipstick-based (lateral flow) format. These give a rapid result (~10-30 minutes) and do not require laboratory equipment and can be performed by clinicians on the ward. They can generally be stored unrefrigerated at 20-25°C for a couple of years.

Available assays
CrAg (cryptococcal antigen)
| ANTIGEN The glucuronoxylomannan (GXM) long branched capsular polysaccharide antigen of Cryptococcus species is released in abundance in body fluids by all common pathogenic species. |
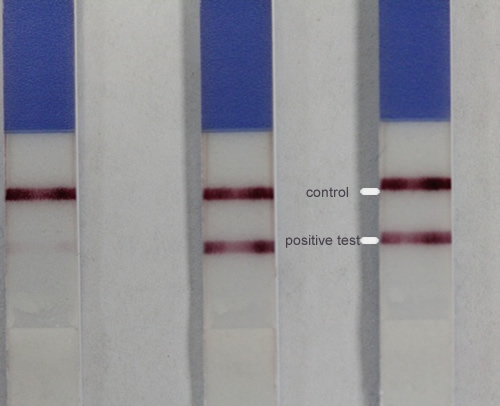
| ASSAY Various detection systems have been abbreviated as CrAg testing. The most widely used has been the latex agglutination test (for serum/CSF). Generally the test gives a positive/negative reading based on a 2-fold dilution of sample; most kits involve boiling and pronase treatment of the serum to avoid the prozone phenomenon and release any bound antigen. Immunoassays may give a quantitative endpoint. More recent LFAs do not require serum pre-treatment, and may allow screening to be performed on other fluids (e.g. urine, BAL). SCREENING: usually done on neat serum/CSF and at 10-fold dilution. TITRE: If screening is positive, a series of 2-fold dilutions are done to measure titre. |
| SUPPLIERS – LATEX AGGLUTINATION: IMMY; Meridian – PRONASE: Meridian – IMMUNOASSAY: IMMY; Meridian; Dynamiker – LFAs (POC): IMMY; Era Biology; Bio-Rad; Liming Bio (StrongStep); Biosynex (CryptoPS) |
| PERFORMANCE All have excellent performance. Read a comparison of latex agglutination kits from Meridian, IMMY, MicroScan and IBL (Tanner et al, 1994). The Biosynex LFA uses a reader to allow the result to be included in the lab IT system and provides slightly higher sensitivity. There are also two bands on the Biosynex and the presence of a higher concentration of antigen in the blood correlates well with the presence of cryptococcal meningitis. – HIV/AIDS: the cryptococcal antigen is positive in CSF in >99% of cases of cryptococcal meningitis and almost all of these patients also have detectable serum antigen positives. Using the new dipstick antigen test, almost all are also positive in urine, although the amount of antigen detectable is 20-fold lower. Serum and possibly urine screening may identify cryptococcal disease early at a less advanced state and may reduce the number of cases of meningitis or the seriousness of the infection. – OTHER IMMUNOCOMPROMISED patients with cryptococcal meningitis, the serum assay is negative in about 30% . CSF titres tend to be lower to those of HIV/AIDS patients with meningitis.No data are published for urine detection. – IMMUNOCOMPETENT patients, with cryptococcal disease (pneumonia or meningitis) the serum CRAG is often negative, and only the CSF or BAL positive, usually at low titre. Often C. neoformans is not recovered from clinical samples, a positive CRAG is the only diagnostic data. |
| CLINICAL USES CrAg titre in CSF is useful for monitoring response to therapy for cryptococcal meningitis. CrAg titre in serum is less useful as titres fall slowly and at different rates in different patients (although a comparison to starting readings can be made). The IMMY assay was shown to give the same results on whole blood as serum or plasma (Williams et al, 2015), which may be useful for screening. |

Aspergillus (galactomannan)
| ANTIGEN Galactomannan is secreted by all pathogenic Aspergillus species, although some isolates are poor producers. A similar metabolite is also produced by several other fungal species including Penicillium spp., Histoplasma capsulatum and other fungi. |
| ASSAY Originally designed for serum, the assay has also been used on BAL, sputum, CSF, pleural fluid, pericardial fluid and tissue. Three ELISA kits and two lateral flow assays (dipsticks) are available for invasive aspergillosis. |
| ELISA CUTOFFS The higher the OD, the greater the likelihood of invasive aspergillosis. The generally accepted cutoff in serum for positives is 0.5 No cutoff has been defined for any other specimen type, but in BAL fluids different authors have recommended cutoffs from 1.0-2.0, with the variation reflecting different patient populations and different dilution factors during BAL fluid collection. Pre-treatment of BAL fluid with Sputasol (dithiothreitol) greatly reduces galactomannan levels. |
| SUPPLIERS – ELISA: BioRad (Platelia); ERA Biology; Dynamiker; FungaDia – LFAs: IMMY (sōna); ISCA Diagnostics (distributed by OLM Diagnostics); Genobio; ERA Biology; Dynamiker. – Pearl Diagnostics (first urine GM test) – -TECO Fast Aspergillus Galactomannan Antigen Lateral Flow Assay – Vircell Aspergillus Galactomannan Ag Virclia |
| PERFORMANCE – ELISA SPECIFICITY: Overall specificity is ~80%. False positives may occur in patients taking β-lactam antibiotics (Tazocin, Augmentin, possibly meropenem, some cephalosporins), as these are produced by fungal fermentation and galactomannan may be carried over during manufacturing. However, false positive results have not been observed with glycopeptides, quinolones or aminoglycosides. In addition, many foods contain galactomannan, e.g. pasta, yoghurt, which can lead to false positives in patients with a permeable small bowel, such as those with mucositis after chemotherapy (Letscher-Bru et al, 1998), this may be more common in children. – ELISA SENSITIVITY: is ~80% in serum among neutropenic patients not receiving prophylaxis; itraconazole and posaconazole antifungal prophylaxis reduce the sensitivity to ~0-20%. In addition, some isolates of Aspergillus appear to be poor producers of galactomannan. While galactomannan is usually detectable up to a week before a CT scan or other standard diagnostic tests are positive, in some cases it is produced late in infection. In non-neutropenic patients, reduced circulating and possibly antigen complexing reduces the serum sensitivity to 0-25%. However, respiratory samples may still be positive. For ventilated intensive care patients with invasive aspergillosis, galactomannan is detectable in ~85% of BAL samples and is the best means currently of establishing a probable diagnosis. Conversely, positive BAL galactomannan tests may be converted to negatives within 3 days by antifungal therapy, so false negative results should be expected soon after anti-Aspergillus therapy is started. A Cochrane review gives a description of how cutoff is related to performance depending on local incidence (de Heer et al, 2019). |
| CLINICAL USES In haematology patients (profound neutropaenia or HSCT), failure of therapy is associated with persistently elevated galactomannan in serum, and conversely a fall in OD by 3 weeks is associated with good outcomes. Galactomannan may be more accurate than BDG in paediatric patients (Singh et al, 2020). |




Histoplasma antigen
| ESSENTIAL The Histoplasma antigen test is included on the 2nd edition of the WHO Essential Diagnostics List for use in patients with advanced HIV/AIDS, and PAHO recommends diagnosis using antigen testing. Disseminated histoplasmosis is an AIDS-defining illness and surpasses tuberculosis as a cause of death among HIV/AIDS patients in Latin America (Cayenne et al, 2018). |
| ANTIGEN Several antigens are detected, including some carbohydrate or glycoprotein structures. The optimal specimen is urine, although blood and BAL fluids have also been tested. |
| ASSAY Mainly enzyme immunoassay. MiraVista has launched a lateral flow (POCT) assay for use with serum and urine. |
| SUPPLIERS – Kits: IMMY; Optimum Imaging – POCT/LFA: MiraVista, OIDx – Submit samples: Miravista |
| PERFORMANCE For a systematic review please see Nacher et al, (2018) – IMMY: sensitivity of 81%, specificity of 99%, diagnostic accuracy of 96%. There is cross-reactivity with Blastomyces dermatitidis, Coccidioides immitis and Paracoccidioides brasiliensis. Read the 510(k) report. – OPTIMUM IMAGING: sensitivity of 95% (100% against culture positives), specificity of 70%, diagnostic accuracy of 86%. Cross-reactivity with Blastomyces dermatitidis, Coccidioides immitis and Cryptococcus neoformans. – MIRAVISTA (sample submission): this assay performs well, with well-characterised specimens (Richer et al, 2016). Antigenuria was detected in 92% of those with disseminated histoplasmosis, and antigenaemia in 100%. Those with severe disease and immunocompromised patients are more often positive. Lower frequencies of positivity are found in those with acute, subacute and chronic pulmonary histoplasmosis. Cross-reactivity was common in patients with blastomycosis. – MIRAVISTA POCT/LFA: sensitivity (96%) and specificity (90%) among culture-positive Colombian patients (Cáceres et al, 2019). |
| CLINICAL USES As Histoplasma capsulatum is slow growing in culture (10 days or more), early diagnosis of disseminated infection requires visualisation of the organism in blood smears, bone marrow, lymph node, skin or lung biopsies. Antigen assays allow earlier diagnosis. |

Candida (mannan)
| ANTIGEN A mannan antigen of Candida albicans circulates in the blood of many patients with candidaemia. It is immunogenic and antibodies are formed over time. |
| ASSAY Latex agglutination |
| SUPPLIERS Kanto Chemical (Cica); Bio-Rad (Platelia); ERA Biology (CLIA); ERA Biology (LFA) |
| PERFORMANCE Several test kits are available with differing performance: – Ramco (Cand-Tec): sensitivity (52.6%), specificity (50.5%), in patients with candidaemia. – Kanto Chemical (Cica): sensitivity (63.2%), specificity (95.5), in patients with candidaemia. – Bio-Rad (Platelia): sensitivity (58%), specificity (93%), with a diagnostic odds ratio (DOR) of 18, in invasive candidiasis, most of whom had (candidaemia). Concurrent detection of anti-mannan antibodies increased the performance of the test to a sensitivity of 83%, specificity of 86%, and DOR of 58. In neutropenic patients, at least one positive mannan or anti-mannan test is positive, and so interpretation is improved by repeat testing and requiring 2 positives for a true positive. The relatively low sensitivity is explained by this antibody complexing. |
| CLINICAL USES Overall diagnostic performance is improved by also measuring anti-mannan antibodies. |
Other antigen tests
| ANTIGEN Tinea unguium antigen kit: DermaQuick A newly developed test for dermatophyte antigen in nails suspected of onychomycosis was released in Japan in 2022. Antigen is usually positive in newly presenting patients with Trichophyton spp. and probably Fusarium spp., and turns negative after successful treatment. As much sub-ungual debris from deep in the proximal lesion should be collected and a nail sample of 1mg or 1mm3 is sufficient for analysis using this antigen test. Its sensitivity is higher than its specificity, using microscopy and culture as the gold standard. The test takes five minutes to deliver an outcome and can be easily read. See Noguchi et al (2023), for more information. |
